These inflammatory phases, which constitute the relapses of multiple sclerosis, follow one another until persistent, chronic inflammation sets in, initiating a cellular cascade that leads to the degeneration of neurons and the onset of permanent disability.
Multiple sclerosis (MS) is an inflammatory disease of the central nervous system that affects the brain, spinal cord and optic nerves, and whose symptoms result from a reduction or even disappearance of communication between certain regions of the brain and spinal cord and peripheral organs.
The nervous system
Via millions of receptors, the nervous system receives sensory information (from the skin, muscles, sense organs), via sensory neurons, about changes (stimuli) coming from the internal environment (the body) or the external environment (the surroundings). The central nervous system (brain and spinal cord) integrates (processes, analyses and interprets) the information it receives: it perceives the nature of the information (source, intensity), compares it with reference values and decides on an appropriate response. The central nervous system reacts to integration by ordering a motor activity (command), via the motor neurons, such as a muscle contraction. The central nervous system controls and receives information from the body’s other organs via the autonomic or vegetative system over which we have no conscious control, such as the heartbeat.
Neurons
Neurons are the cells that transmit information between the central nervous system and the periphery. They are made up of a cell body, an axon and terminal extensions. Axons have the ability to emit and receive electrical signals (nerve impulses), which are the means of sensory and motor communication.
The axons of neurons are protected and nourished by a sheath of lipids, myelin, which acts like insulation around electrical wires. Myelin also accelerates nerve impulses. Myelin forms the white matter of the brain.
Multiple sclerosis lesions
The lesions or “plaques” seen in patients’ brains or spinal cords on magnetic resonance imaging (MRI) are characterised by four components:
- Inflammation, which is the body’s defence reaction to external aggression mediated by immune cells. In multiple sclerosis, the immune reaction is directed against the body itself, known as an autoimmune disease. The immune cells attack a component of the CNS: myelin.
- Demyelination, microscopic observation of a multiple sclerosis lesion. The neurons (cell body and axon) are marked in green, the myelin is marked in red. The protective sheath has disappeared from some axon segments.
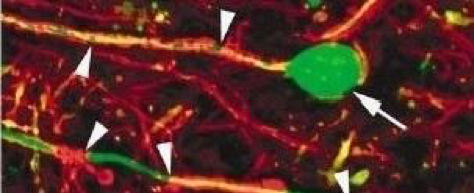
- Neuronal degeneration: deprived of their protective sheath, neurons degenerate and die.
In this microscopy observation, the myelin marked in red has completely disappeared. The axons have retracted and the cell bodies are beginning to die.
This is known as neuropathy.
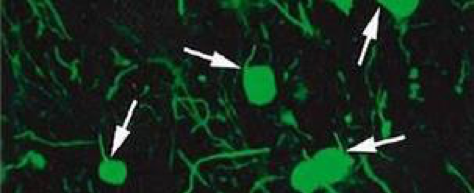
Attack on the myelin and neuronal degeneration
- Myelin repair: in certain lesions, there is a spontaneous phenomenon of myelin repair which, if it occurs at an early stage after the formation of the lesion, prevents the death of neurons and the onset of disability.
This phenomenon is not uncommon, but there is considerable heterogeneity between patients regarding this intrinsic capacity to repair the lesions caused by multiple sclerosis. Nerve impulses disappear in the affected brain regions. Information no longer circulates, and symptoms appear.
At the Paris Brain Institute
- A study conducted by Beatriz Garcia-Diaz and Anne Baron Van Evercooren (DRE INSERM) at the Paris Brain Institute. Brahim NAIT-OUMESMAR and Violetta ZUJOVIC’s team has just identified the mechanism by which Schwann cells, the cells that repair the myelin sheath of peripheral neurons, migrate into the spinal cord.
- Catherine LUBETZKI and Bruno STANKOFF’s team are also conducting projects on the mechanisms of demyelination and demyelination.
- A collaborative study between the 2 teams, led by Céline LOUAPRE and Violetta ZUJOVIC, aims to develop a tool for early assessment of disability progression in order to offer personalised and effective treatment to patients at the start of the disease.
About inflammation
- Several studies by Professor Bruno Stankoff’s team, entitled “Remyelination in multiple sclerosis: from biology to clinical translation“, have revealed new mechanisms of cerebral inflammation in multiple sclerosis, using new imaging tools based on a combination of magnetic resonance imaging (MRI) and positron emission tomography (PET).
- A collaboration between Violetta ZUJOVIC and Fabrizio DE VICO FALLANI has led to the identification of ‘key’ genes that control the ‘pro-inflammatory’ state of macrophages in multiple sclerosis.
About remyelination
- In an article published in the European Journal of Neurology, Vito Ricigliano, Benedetta Bodini and their colleagues at the Brain Institute demonstrate the protective effect of myelin repair on the tissue surrounding lesions in patients with multiple sclerosis. This discovery highlights the potential of new therapeutic strategies and provides new evidence for evaluating the efficacy of remyelinating drugs currently being tested.
- As part of an international consortium, AnneBARON-VAN EVERCOOREN and her colleagues have demonstrated that the remyelination defect present in certain patients with relapsing-remitting multiple sclerosis is not linked to an intrinsic defect in oligodendrocytes, the brain’s myelinating cells, but to the toxic and inflammatory environment of the lesions.
- A study by the “Myelin plasticity and regeneration” team, co-directed by Brahim NAIT-OUMESMAR, has identified new inhibitors of the protein kallikrein 6 (KLK6) and demonstrated their ability to stimulate myelin production in experimental models.
Last updated May 2024.






