Brain mapping consists in finding the brain regions associated with different traits, such as diseases, cognitive functions, or behaviours, and is a major field of research in neuroscience. This approach is based on statistical models and is subject to numerous biases. To try to counter them, researchers from the ARAMIS team, a joint team between the Paris Brain institute and Inria, and their collaborators at the University of Queensland (Australia) and Westlake University (China), propose a new statistical model for brain mapping. The results are published in the Journal of Medical Imaging.
Mapping the brain
Mapping the brain is a challenge that mobilises many neuroscience researchers around the world. The goal of this approach is to identify the brain regions associated with different traits, such as diseases, cognitive scores, or behaviours. This type of study is also known as “Brain-wide association study” and rely on an exhaustive screening of brain regions to identify those associated with the trait of interest.
“The difficulty is that we are looking for a needle in a haystack, except that we don’t know how many needles there are, or in our case, how many brain regions there are to find,” explains Baptiste Couvy-Duchesne (Inria), first author of the study.
Meeting the challenges of signal redundancy
A first challenge lies in the number of brain measurements available per individual, which can quickly reach one million or more. In addition, brain regions are correlated with each other. Some regions are highly connected and associated with many others, like nodes in a network. Others, however, are more isolated, either because they are independent of other brain regions or because they contribute to very specific cognitive trait or brain function.
“If a brain region associated with our trait of interest is part of a highly connected network, the analysis will tend to detect the whole network, because the signal propagates within regions that are correlated with each other,” continues the researcher, “This signal, which may seem very strong at first sight, is in fact redundant. How then can we find the region or regions that really contribute to the trait of interest within the network?”
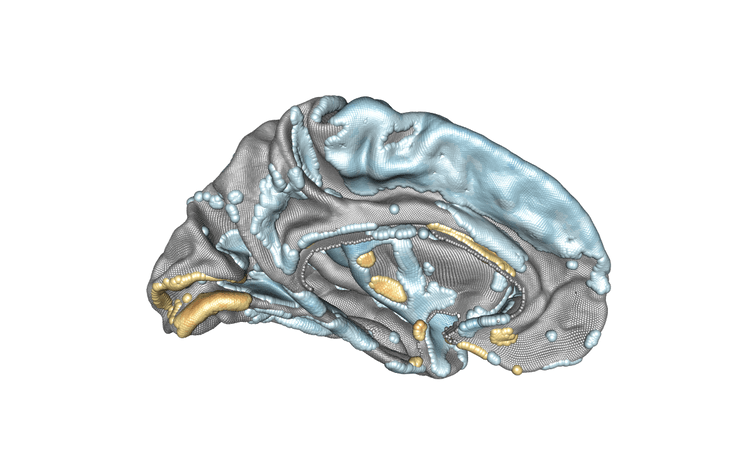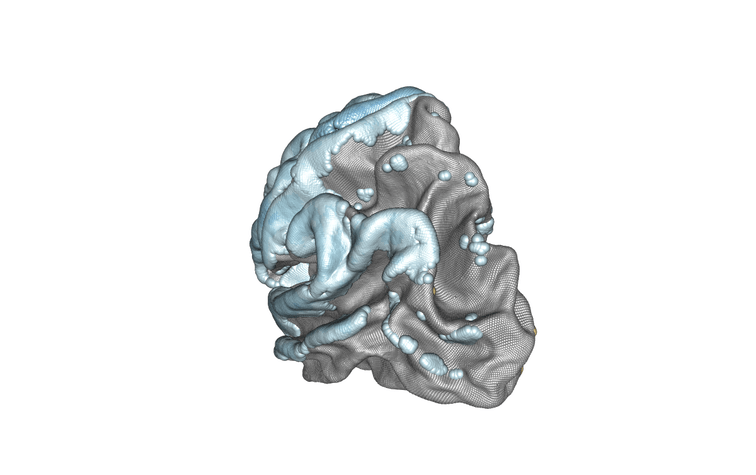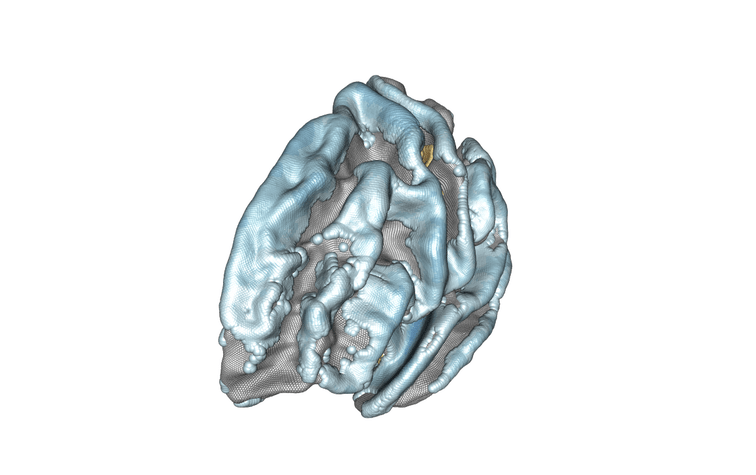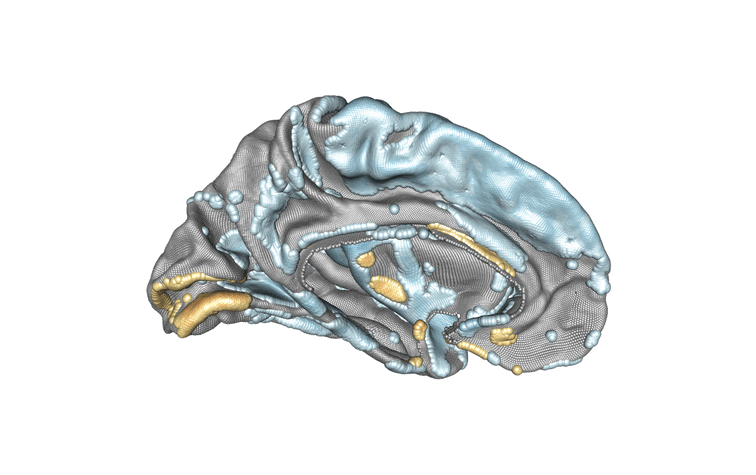
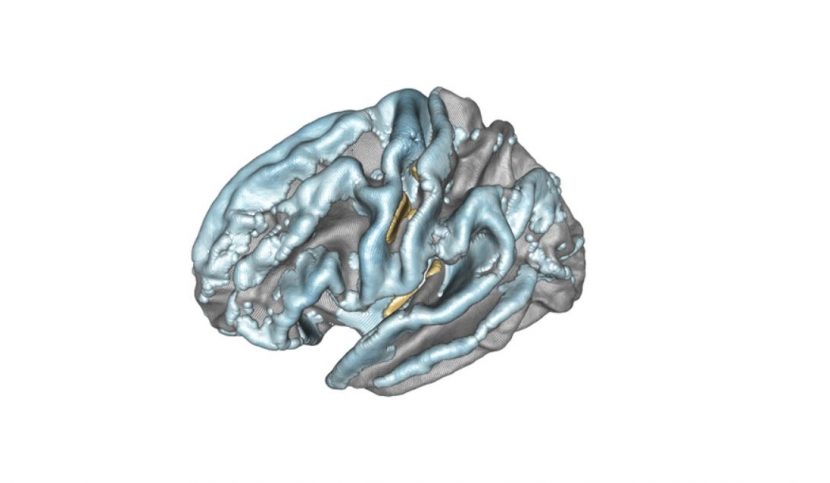
Modelling of the left hemisphere of the brain and the association between subject age and cortical thickness. Positively associated regions are in yellow/orange/red, negatively associated regions are in blue. Credit : Inria/Baptiste Couvy-Duchesne
To solve this problem, the researchers are proposing new statistical methods that are suited to the high dimensional image as well as for modelling the complex correlation structure within the brain.
Simulations to develop new statistical methods
In order to test the developed statistical methods, the researchers need very controlled data. “We cannot compare methods directly on real traits or diseases, since we do not know what we are supposed to find,” explains Baptiste Couvy-Duchesne, “one method could find 10 regions associated with a trait, another 20, although we cannot tell which one is giving the correct answer.”
The key to this solving problem is to use simulations. Researchers use real brain images, but study fake diseases or fake scores, which they have constructed to be associated with dozens or hundreds of predefined brain regions. This way, they are able to check whether the statistical methods detect the expected regions, but also whether they detect others (‘false positives’).
A more robust method and open questions
Once their method had been calibrated through these simulations (which revealed that the proposed approach was more accurate than existing ones) the researchers used real traits as validation.
“Our new method finds fewer regions on average because it manages to remove some of the redundant associations. The next step is to apply it to study Alzheimer’s disease,” concludes the researcher.
A central result of the study it to demonstrate how pervasive are the redundant associations, using the current statistical methods. Thus, many associations identified to date may not be robust of directly pertinent for the trait studied. In addition, several factors that are difficult to control can affect the quality of MRIs, such as head movements or the type of machines used, which can exacerbate the problem and lead to false associations. Beyond the development of more refined analysis methods, the issue of data quality and homogeneity remains crucial.
Source
Baptiste Couvy-Duchesne, Futao Zhang, Kathryn E. Kemper, Julia Sidorenko, Naomi R. Wray, Peter M. Visscher, Olivier Colliot, Jian Yang, “A parsimonious model for mass-univariate vertex-wise analysis,” J. Med. Imag. 9(5), 052404 (2022), doi: 10.1117/1.JMI.9.5.052404.