Vanessa Batista Brochard is CRA coordinator at the Neuroscience Clinical Investigation Center (Neuroscience CIC) at the Pitié- Salpêtrière Hospital headed by Dr Céline LOUAPRE and located within Paris Brain Institute.
CICs were created on the French National Institute of Health and Medical Research’s (Inserm) initiative for the purpose of coordinating studies focusing on Humankind.
The Neuroscience CIC at Paris Brain Institute has staff trained in clinical research for studying neurological and psychiatric diseases under optimal quality, ethics and safety conditions. It can be called on by doctors from the neuroscience Medical-University Department (DMU), by Paris Brain Institute research teams as well as by drug manufacturers.
The Neuroscience CIC operates as a hospital department and currently totals 53 people (doctors, nurses, caregivers, neuropsychologists, laboratory technicians, clinical research associates and administrative staff). It also offers expertise in pharmacology and biostatistics.
Vanessa, what is a CRA’s key role?
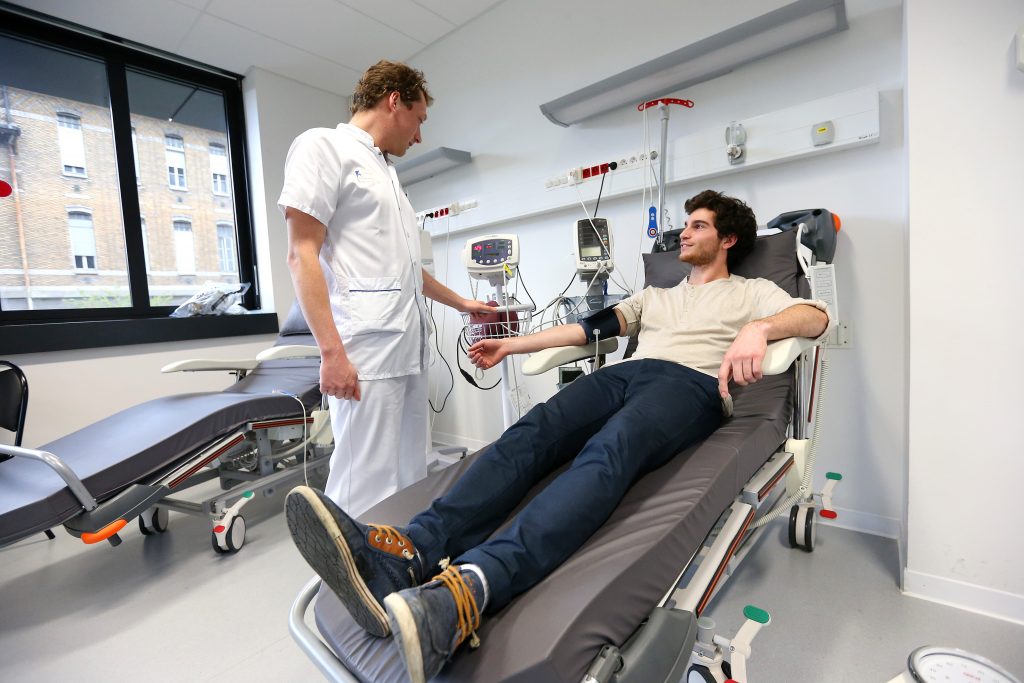
The CRA is pivotal in setting up and running a clinical trial. They provide logistical expertise before, during and after participants are taken in charge. They are the key contact person for the people included in the trial. They contact them, organize the examination schedule and sequencing, explain the purpose of the study to them, make sure they have signed the consent form and that there are no grounds for excluding them from the trial.
©Paris Brain Institute
They accompany the participant throughout the day to the different platforms where the examinations take place and compile and check the clinical and biological data collected during the day.
So, what’s your role as the coordinator?
I’m involved in optimizing research activities as part of the best clinical practices framework. I also sit on the various in-house committees which study the feasibility of the studies in terms of financial, human and technical resources.
On a daily basis, I look after the schedule for a team of 12 clinical research associates. The CRA acts as the interface between the researcher or the industrial, the doctor and the patients.
“The CRA acts as the interface between the researcher or the industrial, the doctor and the patients”, Vanessa Batista Brochard







